Bases
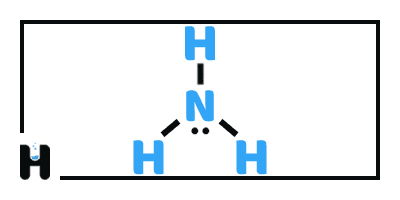
Base is a molecule or an ion which is able to receive a proton (hydrogen ion , H⁺ ) this definition is known as a Bronsted–Lowry base. In basic solution, the pH will be more than 7, and as the strength of alaklinity increases the value of pH will increased.
The second definition is Lewis bases, the bases can chemically donating pairs of electron to an acid. This is considered as a generalization of the Bronsted definition.
Some Examples
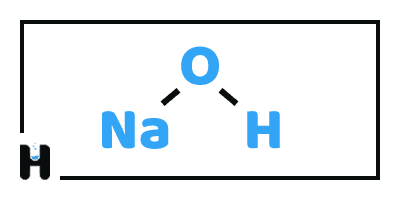
Sodium Hydroxide
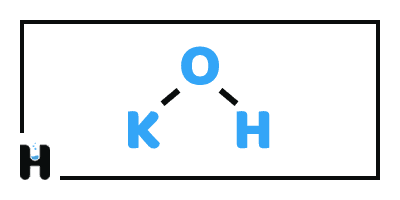
Potassium Hydroxide